This article was published recently in the Australian Journal of
Psychopharmacology.
We thank this journal for permission to publish the seminal work of Dr
Neppe and Dr Young in the area.
Reference:
Neppe VM, Young Z. Buspirone as a new treatment for attention deficit disorder and aggression in children and adolescents. Australian J of Psychopharmacology. 2003, 10, September, 47-56 This is a pioneering article on the use of buspirone use as adjunct to methylphenidate (Ritalin) predominanlty in attention deficit disorder. Based on the data available, buspirone appears an extremely safe medication in adults.
Of course, all patients should be evaluated by a physician.
We gratefully acknowledge the opportunity to publish a minor adaptation of this article which originally appeared in the Australian Journal of Psychopharmacology in 2003. We thank this journal for permission to publish the seminal work of Dr Neppe and Dr Young in the area.
Buspirone is a major medication discussed in Dr Vernon Neppe's classic sciction book, Cry the Beloved Mind: A Voyage of Hope
See Also:
KEY WORDS: Buspirone, attention deficit hyperactivity disorder (ADHD), aggression, irritability, impulsivity, serotonin 1A
ABSTRACT
ADHD patients often have problems linked with difficulty concentrating /distractibility and an impulsivity manifesting as irritability and low frustration tolerance. Because many patients are not well controlled using psychostimulants, adjunctive therapies have been tried with limited success or potential accentuation of side-effects. A significant ADHD symptom that is commonly poorly controlled with psychostimulants is irritability and impulsivity. In a clinical pilot study with retrospective review, and blind rankings of patient response, buspirone adjunct (30mg/day median) to psychostimulants improved ADHD in the young (N=39, P<0.0001) and aggressive non-ADHD children (N=22, p<0.01). Buspirone clinically improved several target symptoms of focus (p<0.0001); irritability/ aggression/ impulsivity (p<0.00001); sleep disturbance (p<0.001); anxiety/ depression/ lability (p<0.01), and hyperactivity (p<0.01). Clinical improvements occurred in more than 90% of ADHD and 80% of non-ADHD patients. Buspirone was well tolerated with few side-effects. The specificity of buspirone for serotonin 1A receptors suggests that the therapeutic effects are modulated through this receptor. Improvement of inattention and impulsivity / irritability symptoms but not hyperactivity suggests that the former symptoms are linked at a neurotransmitter level. The implications are that current thinking linking impulsivity/ irritability with hyperactivity may not be correct.
BACKGROUND
Attention Deficit Hyperactivity Disorder Key Elements
Attention deficit hyperactivity disorder (ADHD) often presents in early or middle childhood with school or behavioural problems linked to difficulty concentrating and distractibility. Hyperactivity, impulsivity, irritability and low frustration tolerance are common. A quarter of patients have subtle learning disabilities, and disputably up to half the ADHD patients have emotional problems which may be primary or secondary to the ADHD, leading to a heterogeneity in the diagnosis of the condition. Epidemiological studies suggest that 3%-5% of all children have ADHD with 75%-90% being male. (Brown, 1991; Farason & Ford, 1994; Jacobvitz, Sroufe, Stewart & Leffert, 1990)
The two key groups of symptoms in ADHD are inattention (distractibility and concentration disturbance) and hyperactivity / impulsivity (increased activity and the impulsivity/irritability/aggression continuum) (APA, 2000). Consequently, the major presenting features of the ADHD spectrum are either impairments of school performance or conduct disorder. These features invariably manifest themselves in both the contexts of school and home environments for a prolonged period, generally years. (APA, 1987; 1994). There are no specific physical features associated with ADHD. Genetically, ADHD may be linked with a deficiency of a non-specific gene variant of the Dopamine D2 receptor gene, which is also common in Tourettes syndrome and alcoholism.
Current pharmacological treatments for ADHD
Psychostimulants are the most common treatments for ADHD but are not benign agents. Psychostimulants (methylphenidate, pemoline, dextro-amphetamine) work well for the attention and hyperactivity features, but possibly less effective for the anger and impulsivity. Their mechanism of action is poorly understood, but has been linked with paradoxical" effects on the reticular activating system (Jacobvitz et al, 1990; Trent, Nakamura & Tepper, 1991). Psychostimulants have side-effects such as tics and arguably dependence. Such addiction may be less in the ADHD population than potential abuse by other family members (Ahmann, Waltonen, Olson, Theye, Van & Laplant, 1993; Elia, Borcherding, Rapoport & Keysor, 1991; Johnson, Handen, Lubetsky & Sacco, 1994; Klorman, Brumaghim, Fitzpatrick & Borgstedt, 1991; Murphy, Pelham & Lang, 1992; Rapport, Denney, Dupaul & Gardner, 1994; Warneke, 1990). When used as sole pharmacotherapy, in apparently optimised doses, psychostimulants achieve good control in only about half the cases.
This limited efficacy lead to a need to use unapproved adjunctive interventions such as tricyclic antidepressants, phenothiazines or clonidine (Schvehla, Mandoki & Sumner, 1994; Hart & Hart, 1992). None of the adjunctive drugs have been adequately researched, but there are anecdotal reports of their usage. Other adjuncts used have included fluoxetine venlafaxine and bupropion (Yu, Smith, Smith & Lyness, 1986). Fluoxetine initially has appetite suppression effects (Barrickman, Noyes, Kuperman, Schumacher & Verda, 1991). All three antidepressants may act like stimulants in ADHD (Barrickman, Perry & Allen, 1995). They may be more useful for particular conditions co-morbid with ADHD, such as anxiety, depression or OCD, than the core symptoms of the disorder (Brown, 1991). It has been found that these adjuncts treatments do not maintain initial effects beyond several months, and are associated with side-effects. They may be regarded as innovative pharmacotherapies as they go beyond regulatory guidelines (Neppe, 1990; Ratey, Greenberg & Lindem, 1991), reflecting a new phase of using adjuncts to psychostimulant medication as part of the management of these disorders. The utilization of a safe, effective adjunctive agent, with a theoretical basis for that effectiveness and empirical guidelines for its use would be valuable clinically. A significant ADHD symptom that is poorly controlled with psychostimulants is irritability (Murphy, et al, 1992).
Serotonin, serotonin 1A, buspirone and irritability
Evidence exists for serotonin receptor involvement in the aggression spectrum from animal models as well as human studies with a particular role for serotonin 1A receptors (Bonson, Johnson, Fiorella, Rabin & Winter, 1994; Moeller, Steinberg, Petty, et al, 1994; Olivier & Mos, 1992; McMillen, Chamberlain & Davanzo, 1988; Neppe, 1993; Bonson, et al, 1994; McMillen, et al, 1988; McMillen, Scott, Williams & Sanghera, 1987; Ratey, et al, 1992; Stanislav, et al, 1994; Olivier, et al, 1992; Colella, Ratey & Glaser, 1992; Neppe, 1993; Gedye, 1991). Furthermore this receptor is strongly linked with irritability and impulsivity (Neppe, 1999a).
Buspirone is a serotonin 1A partial agonist drug post-synaptically, and a pure agonist at the autoreceptor level (Neppe, 1990). From a theoretical point of view it is not surprising that it has been demonstrated to improve adults with anger and irritability (Neppe, 1999a). Consequently, if buspirone is helpful in the poorly controlled, psychostimulant treated ADHD children with residual irritability and impulsivity, would imply an effect mediated through serotonin 1A neurotransmission, a new theoretical idea in ADHD. Moreover, if non-ADHD children with temper tantrums receiving buspirone as monotherapy or as an adjunct to drugs other than psychostimulants respond, then this would support an action of the buspirone independent of any psychostimulant influence on the serotonin 1A receptor. Furthermore if the buspirone also assisted with concentration disturbance in the ADHD psychostimulant treated group, it would support serotonin 1A involvement in concentration. Secondly, if the buspirone did not help hyperactivity, then the ADHD hyperactivity is unlikely to be linked with serotonin 1A. Thus the notion of linking impulsivity / irritability with hyperactivity, which occurs in the DSM-III and DSM-IV-TR nomenclature is incorrect. Instead, the impulsivity / irritability may be more closely linked biochemically with the inattention spectrum.
STUDY DESIGN
Rationale
Buspirone, in the adult population is a safe drug (Neppe, 1999b), but, there is little research in children only case reports (Holttum, Lubetsky, & Eastman, 1994; Zwier & Rao, 1994; Leonard, Topol, Bukstein, Hindmarsh, Allen & Swedo, 1994; Stanislav, Fabre & Crismon, 1994; Ricketts, Goza & Ellis, et al, 1994; Soni & Weintraub, 1992; Realmuto, August & Garfinkel, 1989; Alessi & Bos, 1991; Kranzler, 1988; McCormick, Rizzuto & Knuckles, 1994; Balon, 1990). The anxioselective effects of buspirone take three or four weeks to fully manifest (Neppe, 1990; Neppe, 1999 b). It has early effect on irritability and concentration in the anxious patient. The use of buspirone in ADHD was prompted by its anti-aggressive effects combined with the effects noted on irritability and concentration in other conditions. As an adjunct in children and adolescents who had not responded to psychostimulants it was hypothesized that buspirone would be effective.
The population of patients was children and adults prescribed methylphenidate, pemoline or rarely amphetamines. No adequate literature has appeared in this area. There are two case reports using buspirone in ADHD: One is of a child with ADHD plus aggression exists in an inpatient receiving 45mg buspirone per day when the aggression not the ADHD improved (Quiason, Ward & Kitchen, 1991), the second an adult ADHD patient (Balon, 1990). A small double-blind, placebo controlled, crossover study, gave 5 mg buspirone twice a day on weekdays for two weeks (McCormick, Rizzuto & Knuckles, 1994). There was a response in nine of the subjects studied.
Hypotheses
1. Buspirone adjunct to psychostimulants in ADHD (with or without a second diagnosis) will improve the patients condition.
2. Buspirone alone or as adjunct to other medications will improve the condition of children with anger problems.
3. Buspirone will prove to be safe.
4. Buspirone will improve ADHD ratings in addition to target symptoms of irritability, concentration, sleep, hyperactivity, emotionality and somatic features.
METHODS
Setting
The study was a retrospective case analysis of the clinical data derived from patients at a Child and Adolescent Center under the supervision of Dr. Young. All patients and their families consented to buspirone as adjunct to psychostimulants (Young, 1992).
Sample
Patients were aged 5 to 17 years.
Patients conformed to basic follow- up criteria:
1. At least three follow up sessions with medication compliance.
2. Treatment with buspirone.
3. Absence of mental retardation or disabling physical condition requiring concomitant medication for physical disease.
4. Duration of treatment evaluation of at least 6 months.
5. The sample for analysis was restricted to the end date set for the original research analysis and constituted the first forty ADHD patients being treated with buspirone as well as a group of non-ADHD patients who had presented by that time who conformed to the criteria.
The original sample consisted of ADHD patients who had irritability as a target feature. Later, patients who had not responded well with sleep disturbance and / or anxiety were also added. The ADHD population was divided into two as a substantial proportion of children referred to clinics with ADHD have comorbid diagnoses. (APA, 1987): pure ADHD patients and ADHD plus a second diagnosis.To establish if our effect were purely on ADHD patients, we added another target group of non-ADHD children with temper tantrums and a non-ADHD group who did not exhibit aggression.
Data evaluation
Subclassification of symptoms.
The data was examined retrospectively based on the clinical comments in the chart and any completed forms. Feedback on particular symptoms when present or relevant was provided by the treating psychiatrist (ZOY). Patients were also scored retrospectively by the evaluator (VMN) according to a scale of improved or not improved and statistically analysed. Not improved was separated into "Remains the same" or "Got worse".
Ratings were based on:
1. Overall general symptom response
2a. irritability/ impulsivity as one cluster named "Aggression,"
2b. sleep as a second cluster,
2c. hyperactivity as a third measure,
2d. focus and finally,
2e. the variable of mood-related symptoms - anxiety, depression, and mood lability - called adl
Additionally, a listing was made for sm implying somatic symptoms but the total sample size across all groups was too small (3) for statistical analysis. Further explanation of the assessment of symptoms is provided in Table 1.
Symptoms were rated as 1 or w (worse), 0 (same), or +1 (better). No attempt at further ordinal ranking was made, because it was difficult to separate mild and significant improvement. The rankings were dependent on all information pertaining to change that was available, but more often based on the family's impression of progress, although at times, there were school-related performance aspects.
Statistical analysis
Results were clustered into two groups - improved and not improved (combining the unchanged and deteriorated groups) and Fishers exact test was applied to the one tailed hypothesis of the patients improving on buspirone. As there was no control group, a stringent criterion of response, namely that 50% would respond was assumed. This diminished the power of the statistical results but allowed a more conservative comparison.
RESULTS
There were 69 patients, three dropped out (1 for non-compliance, 2 for failed follow-up) producing a final sample size of 66. Because of the open, clinical nature of the study, statistics have limited meaning but relevant clinical improvements occurred in > 90% of ADHD and 80% of non-ADHD patients. The combined initial sample size was broken down to: 40 ADHD less one dropout; 26 pure ADHD, 14 ADHD including 1 dropout. The Non ADHD group included 23 with aggression, 4 without aggression and 2 dropouts making 29.
The ADHD group, as expected, was predominantly male, and the non-ADHD group was predominantly female with more heterogeneous diagnoses. The youngest patient was aged 5.5 years. Demographic data for the sample is outlined in Table 2.
Average and median doses of buspirone approximated 30 mg per day in all the groups. The buspirone was almost always prescribed as 10mg TID. Adjunctive buspirone was used with the consequence that all ADHD patients and ADHD-plus group were receiving psychostimulants, predominantly methylphenidate, in doses of 30 mg per day. The non-ADHD group had a majority of patients on no psychotropic medications other than buspirone (22 of 29 originally, or 20 of 27 after dropouts). The target symptoms in the non-ADHD group were predominantly aggression and irritability occurring in 23 of the 27 for analysis, 25 of the 29 initially. The four patients without ADHD receiving buspirone for other reasons had their data separated out.
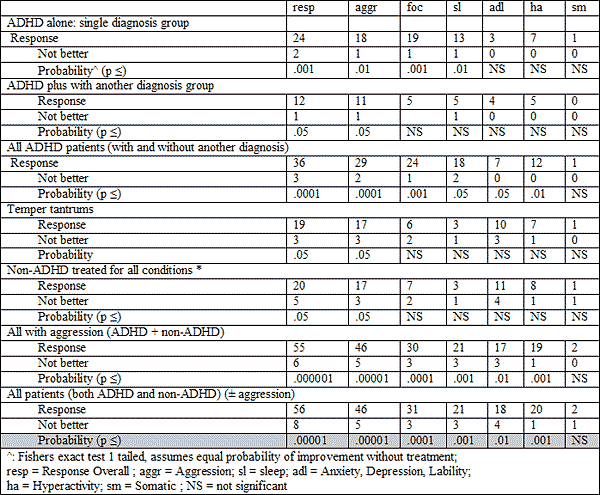
Several target symptoms in the ADHD group improved: (Table 3):
1. Focus: concentration/ distractibility / school functioning: Of those who still had concentration disturbance, 24 improved out of 25 in the ADHD groups and 7 of 9 in the non-ADHD group.
2. Aggression: irritability/ anger/ temper tantrums/ low frustration tolerance: Overall >90% improvement combining both groups (p< 0.00001).
3. Improved sleep (p<.001).
4. Improved anxiety or anxietydepression. (p< 0.01).
These results are also clear when the ADHD group is separated into two. Due to small samples sizes the statistical tests lack power.
5. Hyperactivity improved in all 12 patients who had this as a residual pre-buspirone symptom.
A further population of aggressive non-ADHD children also responded well to buspirone alone in similar doses.
Side-effects
Overall, side-effects were reported rarely - dizziness in 2.9%, headache, nausea and sleepiness in 1.5% each. Overall, the incidence of patients with side-effects attributable to the buspirone was 5.8% given that the patient with headache also had dizziness. A non-buspirone related side-effect was attributed to thioridazine which when added to one 16 year old female patient, who became hypomanic.
Discussion
The results of the study need to be interpreted cautiously. Buspirone appears to be clinically effective as an adjunctive treatment in attention deficit disorder children who have incompletely responded to psychostimulants. Buspirone is versatile in that it can be used in classical ADHD or when ADHD coexists with other diagnoses. Buspirone was also effective in children with temper tantrums without attention deficit disorder.
The limitations of the study need to be recognized. This was not a randomised placebo controlled study, but instead a case series of retrospective data with possible biases by the treating clinicians and consequent compromised rating biases. Clinical limitations of this study included: inadequate checks of compliance; variable follow-up; evaluations limited in scope; histories based on reports given from members of family; an inconsistent amount of data existing with each patient; other medications confounding evaluations. The population of ADHD children all had incompletely responded to psychostimulants, while those who had deteriorated on medication were not included in the chart review.
Although DSM III-R criteria for diagnosis were used, the patient population appears in all cases to have met the later DSM 1V-TR criteria for either a Combined or Predominantly Hyperactive / Impulsive subtype of this condition. Most patients with ADHD had Connors 10 point scores of 20 or more reflecting moderate to severe ADHD; similarly, the non-ADHD group was generally impaired and their other condition was often marked. None of the patient population was mentally retarded. These features further add to the bias of this population and the results may not necessarily generalize to mild ADHD or mildly impaired non-ADHD children.
Finally, the non-ADHD group was heterogeneous. Nine had post-traumatic stress disorder as did nine of the ADHD group; five non-ADHDs had conditions like Pervasive Developmental Disorder. Five others had borderline tendencies, five more had adjustment difficulties, two were labeled schizo-affective and three had affective illness. Whereas 20 of 27 took only buspirone, others were on medications such as lithium, valproate and neuroleptics, and in three instances, on combinations.
Hyperactivity improved in all 12 patients who still had this as a residual pre-buspirone symptom. This was an unexpected finding. In part, this may have related to operational interpretations. However, given that the hyperactivity still existed after psychostimulant, it is possible this was not the typical hyperactivity encountered in ADHD. Some support comes from an anecdotal group where we have found no improvement in hyperactivity when we gave buspirone alone in 5 cases. Further clinical study is warranted, as the consequences would suggest a functional limitation or extension for the serotonin 1A receptor.
The quantification of phenomena pertaining to impulsive behaviours and aggression spectrum behaviour ranging from anger to violence to rage to dyscontrol has always been difficult. The interpretation of changes with these patients was not easy. However, clinically significant improvements were seen. Given the data in adults, it is likely buspirone does indeed improve irritability in this population. The anger components were almost invariable in our subpopulation involving three quarters of the ADHD population and four-fifths of the non-ADHD group. The aggression response is often marked.
Initially, the resulting improvement of sleep from our data may be surprising given the non-sedative properties of buspirone in the adult and animal population as well as in these patients (only one complained of sedation). However, this can be seen in the context of the psychophysiological re-regulation over time and it appears to be a symptom that improves more over weeks than over days.
Almost one third of all the children (and one sixth of the ADHD groups) had anxiety, or Mixed Anxiety Depression. As anticipated by its indication in adults, buspirone induced significant improvement in more than four-fifths of patients. In addition, such symptoms as concentration improved in all populations when the abnormalities were present initially. As with aggression, it is possible such improvement is a direct serotonin 1A related effect.
We cannot interpret accurately data as to the onset of improvement of the target symptoms with these patients. In general, patients appeared more improved a month after initiation of buspirone than a week later. Early target symptoms of response anecdotally related to the improvement in irritability, impulsivity and calming elements. Evaluations were based on the interpretation of the treating clinician. Information was frequently assisted by outside information such as scorings of reports by members of family of changes in the target symptomsalthough these have not been specifically quantitated, the improvements frequently were greater at Week 4 (e.g. a lot) compared with Weeks 1 and 2, where changes were reported as a little. Such rankings were based on the feedback of the patients, families, relationships and ability to perform daily tasks.
Buspirone was well tolerated. The absence of drop-outs due to side-effects is remarkable. These children and adolescents apparently showed an even better tolerance than is reported in the adult population. Nausea occurred in only one patient who was non-compliant and was dropped from the study. This is a relatively common effect with serotonin active drugs. In adults it occurs at a rate of 5-10% above placebo with buspirone. Dizziness occurred in less than 5% of the sample, fewer than a clinical incidence in adults estimated at 12% to 40% in higher doses. Headache in the child population was reported once with the dizziness in the same patient, again less than the incidence in adults estimated at 5%-10% above placebo. Dizziness with buspirone termed non-vertiginous dizziness (Neppe, 1993; Neppe, 1999b) is by far the most common side effect in adults, and it apparently can be used as an index of dosage tolerance. Headache is seen with buspirone in adults, and often seems to be an alternative for non-vertiginous dizziness but is uncommon.
In children, dosage appears similar to adults beginning at 5mg three times daily unless the child has low body mass (e.g. < 65 lbs), or young (e.g. 7 or less). The dose is built up by 5mg every three days, although in this sample, 30mg per day was achieved even quicker. Subsequent to this study, doses of 5mg TID instead of 10mg TID may be more appropriate in the absence of anxiety / depression symptoms and in the presence of irritability. There are still limited data to back up this clinical impression.
In this study, no major attempts were made to modify the dose of psychostimulant. This was a clinical series of patients and given that they had improved, they were, in general, maintained on the same dose of medication. There are no adequate data to indicate if diminution of dose is possible, or for that matter whether the buspirone dose can be diminished. If methylphenidate or pemoline dose can be decreased, this should limit the potential long and short-term side effects, and the buspirone may prove a safer alternative.
Biochemically, the neurotransmitter dopamine is regarded as important in ADHD. (Kostrezewa, Brus, Kalbfleisch, Perry & Fuller, 1994). Whereas this is clearly true, the current research suggests a role for the serotonin 1A receptor based on the selectivity of buspirone. Based on current theoretical data (Neppe, 1993), it can be suggested that buspirone action is on the serotonin 1A receptor in the context of irritability and impulsivity, and likely in the attentional sphere. Given the common response to buspirone of distractibility and impulsivity / irritability but lack of effect on hyperactivity as a target symptom, we hypothesize that the irritability / impulsivity spectrum is pharmacologically closer to distractibility than to hyperactivity.
Most of the limited responsive patients with ADHD have the hypothesized serotonin 1A cluster of irritability, temper tantrums, frustration, impulsivity and immediate gratification cluster. We believe it is unlikely the anti-anxiety effect would be the only mechanism of action of buspirone in ADHD because the conventional anxiolytics which act almost immediately in anxiety, such as the benzodiazepines and barbiturates, anecdotally make ADHD (Fargason, et al, 1994) or autism (Marrosu, Marrosu, Rachel & Biggio, 1987) worse as the hyperactivity becomes aggravated.
As a clinical impression, buspirone has not shown loss of efficacy in the lengthy follow-up cases. This may be a critical finding because reports of loss of efficacy of some SSRIs in a subpopulation of depressed patients treated for 6 months or a year are emerging. Clinically, buspirone does not appear to lose efficacy in anxiety or mixed anxiety depression either, possibly because partial agonism compensates for distortions in overall changed regulation of serotonin.
Further directions
A double blind study of buspirone versus placebo as psychostimulant adjunct is now necessary. Consideration may be given to comparison of buspirone versus other medications such as clonidine or bupropion as adjunctive therapy in this subpopulation of ADHD. In addition, buspirone alone should be studied in double blind fashion in children and adolescents without ADHD but with irritability or anxiety syndromes. An open study of buspirone in children with attention deficit disorder has been performed (Malhotra & Santosh, 1998), but there have been no double blind studies.
TABLE 1: CRITERIA FOR SYMPTOM RATING
|
Impulsivity / Irritability or Aggression
Associated features of ADHD are recognized in DSM IV-TR and vary depending on age and developmental stage. They may include
low frustration tolerance, temper outbursts, bossiness, stubbornness, excessive and frequent insistence that requests be met.
These are not regarded as primary features of the ADHD syndrome despite their frequency. Temper tantrums were included in this symptom set.
The operational criteria for differentiating impulsive behaviour under aggression or hyperactivity included the following: hyperactivity
required one of the DSM IV criteria or the impulsivity symptom of blurting out answers; inability to delay gratification was classified under irritability.
Other symptoms operationalized
a,d,l group : mood lability, demoralization, dysphoria, rejection by peers, and poor self-esteem are also associated features of ADHD are
recognized in DSM IV and 111R. These as well as anxiety and depression constituted the a,d,l group.
focus was reflected by calming, better school performance academically, ability to maintain attention and lesser distractibility,
sleep disturbance of any kind, based on any sleep disturbance, initial paroxysmal or terminal insomnia,
hyperactivity as a third measure, including fidgetiness and pacing and restlessness, on changes in terms of calmness and concentration and
attentional and school performance behaviour as the attentional variable, and finally, on the variable of mood-related symptoms - anxiety,
depression, and mood lability.
Side-effects
When specifically elicited or reported, side-effects were recorded.
|
TABLE 2: DEMOGRAPHIC FEATURES OF THE POPULATION
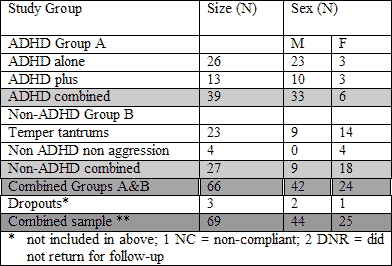
TABLE 3: SYMPTOM RESPONSE SUMMARY AFTER BUSPIRONE TREATMENT
REFERENCES
AHMANN PA, WALTONEN SJ,
OLSON KA, THEYE FW, VAN EAJ, LAPLANT RJ (1993) Placebo-controlled evaluation of
Ritalin side effects. Pediatrics, 91 (6):1101-6
ALESSI N, BOS T (1991)
Buspirone augmentation of fluoxetine in a depressed child with
obsessive-compulsive disorder. American Journal of Psychiatry, 148
(11):1605-1606
AMERICAN PSYCHIATRIC
ASSOCIATION COMMITTEE (2000). Diagnostic and Statistical Manual: DSM-1V-TR,
Four Text Revision, Washington,
D.C.: American Psychiatric
Association
AMERICAN PSYCHIATRIC
ASSOCIATION COMMITTEE. (1987) Diagnostic and Statistical Manual: DSM 111 R,
Third (revised), Washington,
D.C: American Psychiatric Association
BALON R (1990) Buspirone for
attention deficit hyperactivity disorder? Journal of Clinical
Psychopharmacology, 10 (1):77
BARRICKMAN L, NOYES R,
KUPERMAN S, SCHUMACHER E, VERDA M (1991) Treatment of ADHD with fluoxetine: a
preliminary trial. Journal of the American
Academy of Child and
Adolescent Psychiatry, 30 (5):762-7
BARRICKMAN LL, PERRY PJ,
ALLEN AJ, et al. (1995) Bupropion versus methylphenidate in the treatment of
attention-deficit hyperactivity disorder. Journal of the American Academy
of Child and Adolescent Psychiatry, 34 (5):649-57
BONSON KR, JOHNSON RG,
FIORELLA D, RABIN RA, WINTER JC (1994) Serotonergic control of androgen-induced
dominance. Pharmacology Biochemistry and Behavior 49 (2):313-22
BROWN CS (1991) Treatment of
attention deficit hyperactivity disorder: a critical review. DICP, 25
(11):1207-13
COLELLA RF, RATEY JJ, GLASER
AI (1992) Perimenstrual aggression in mentally retarded adult ameliorated by
buspirone. International Journal of Psychiatry in Medicine, 22(4):351-6
ELIA J, BORCHERDING BG,
RAPOPORT JL, KEYSOR CS (1991) Methylphenidate and dextroamphetamine treatments
of hyperactivity: are there true nonresponders? Psychiatry Research, 36
(2):141-55
FARGASON RE, FORD CV (1994)
Attention deficit hyperactivity disorder in adults: diagnosis, treatment, and
prognosis. Southern Medicine Journal, 87 (3):302-9
GEDYE A (1991) Buspirone
alone or with serotonergic diet reduced aggression in a developmentally
disabled adult. Biological Psychiatry, 30 (1):88-91
HART SD, HART LL (1992)
Clonidine in attention deficit hyperactivity disorder. Annals of
Pharmacotherapy, 26 (1):37-9
HOLTTUM JR, LUBETSKY MJ,
EASTMAN LE (1994) Comprehensive management of trichotillomania in a young
autistic girl [see comments]. Journal of the American Academy
of Child and Adolescent Psychiatry, 33 (4):577-81
JACOBVITZ D, SROUFE LA,
STEWART M, LEFFERT N (1990) Treatment of attentional and hyperactivity problems
in children with sympathomimetic drugs: a comprehensive review. Journal of the American Academy of Child and Adolescent
Psychiatry, 29 (5):677-88, 1990
JOHNSON CR, HANDEN BL,
LUBETSKY MJ, SACCO KA (1994) Efficacy of methylphenidate and behavioral
intervention on classroom behavior in children with ADHD and mental
retardation. Behavior Modification, 18 (4):470-87
KLORMAN R, BRUMAGHIM JT,
FITZPATRICK PA, BORGSTEDT AD (1991) Methylphenidate speeds evaluation processes
of attention deficit disorder adolescents during a continuous performance test.
Journal of Abnormal Child Psychology, 19 (3):263-83
KOSTRZEWA RM, BRUS R,
KALBFLEISCH JH, PERRY KW, FULLER RW (1994) Proposed animal model of attention
deficit hyperactivity disorder. Brain Research Bulletin, 34 (2):161-7
KRANZLER HR (1988) Use of
buspirone in an adolescent with overanxious disorder. Journal of the American Academy of Child and Adolescent
Psychiatry, 27 (6):789-90
LEONARD HL, TOPOL D,
BUKSTEIN O, HINDMARSH D, ALLEN AJ, SWEDO SE (1994) Clonazepam as an augmenting
agent in the treatment of childhood-onset obsessive-compulsive disorder.
Journal of the American
Academy of Child and
Adolescent Psychiatry; 33 (6):792-4
MALHOTRA S., SANTOSH P. J.
(1998) An open clinical trial of buspirone in children with
attention-deficit/hyperactivity disorder. Journal of the American Academy
of Child and Adolescent Psychiatry, 37 (4): 364-37
MARROSU F, MARROSU G, RACHEL
MG, BIGGIO G (1987) Paradoxical reactions elicited by diazepam in children with
classic autism. Functional Neurology, 2 (3):355-61
MOELLER FG, STEINBERG JL,
PETTY F, et al. (1994) Serotonin and impulsive/aggressive behavior in cocaine
dependent subjects. Progress in Neuropsychopharmacology and Biological
Psychiatry, 18 (6):1027-35
MURPHY DA, PELHAM WE, LANG AR
(1992) Aggression in boys with attention deficit-hyperactivity disorder:
methylphenidate effects on naturalistically observed aggression, response to
provocation, and social information processing. Journal of Abnormal Child
Psychology, 20 (5):451-66
McCORMICK LH, RIZZUTO GT,
KNUCKLES HB (1994) A pilot study of buspirone in attention deficit
hyperactivity disorder. Archives of Family Medicine, 3 (1):68-70
McMILLEN BA, CHAMBERLAIN JK,
DAVANZO JP (1988) Effects of housing and muricidal behavior on serotonergic
receptors and interactions with novel anxiolytic drugs. Journal of Neural
Transmission, 71 (2):123-32
McMILLEN BA, SCOTT SM,
WILLIAMS HL, SANGHERA MK (1987) Effects of gepirone, an aryl-piperazine
anxiolytic drug, on aggressive behavior and brain monoaminergic
neurotransmission. Naunyn Schmiedeberg's Archive of Pharmacology 335 (4):454-64
NEPPE VM.(1990) Innovative
Psychopharmacotherapy, Revised 1st edit (1st edition 1989), New York: Raven Press, 1-225 & i-xi
NEPPE VM (1990) Buspirone:
an anxioselective neuromodulator. In: Innovative Psychopharmacotherapy, Revised
1st edition,. Neppe VM ed. New York:
Raven Press, 35-57, Ch 2
NEPPE VM. (1993) Serotonin
1A neuromodulators clinical implications for the elderly. In:
Psychopharmacology in Old Age. Bergener M, Belmaker RH, Tropper MS ed. New York: Springer
Verlag, 222-238
NEPPE VM (1999a) The
serotonin 1A neuromodulation of aggression : Bimodal buspirone dosage as a
prototype anti-irritability agent in adults. Australian J of
Psychopharmacology. 9: 8-25.
NEPPE VM (1999b) Cry the
Beloved Mind: A Voyage of Hope. Seattle:
Brainquest Press DBA Brainvoyage.com, Ch 9-11.
OLIVIER B, MOS J (1992)
Rodent models of aggressive behavior and serotonergic drugs. Progress in
Neuropsychopharmacology and Biological Psychiatry, 16 (6):847-70
QUIASON N, WARD D, KITCHEN T
(1991) Buspirone for aggression. Journal of the American Academy
of Child and Adolescent Psychiatry, 30 (6):1026
RAPPORT MD, DENNEY C, DUPAUL
GJ, GARDNER MJ (1994) Attention deficit disorder and methylphenidate:
normalization rates, clinical effectiveness, and response prediction in 76
children. Journal of the American
Academy of Child and
Adolescent Psychiatry, 33 (6):882-93
RATEY JJ, GREENBERG MS,
LINDEM KJ (1991) Combination of treatments for attention deficit hyperactivity
disorder in adults. Journal of Nervous and Mental Disease, 179 (11):699-701
RATEY JJ, LEVERONI CL,
MILLER AC, KOMRY V, GAFFAR K (1992) Low-dose buspirone to treat agitation and
maladaptive behavior in brain-injured patients: two case reports [letter].
Journal of Clinical Psychopharmacology, 12 (5):362-4
REALMUTO GM, AUGUST GJ,
GARFINKEL BD (1989) Clinical effect of buspirone in autistic children. Journal
of Clinical Psychopharmacology, 9 (2):122-5
RICKETTS RW, GOZA AB, ELLIS
CR, et al. (1994) Clinical effects of buspirone on intractable self-injury in
adults with mental retardation. Journal of the American Academy
of Child and Adolescent Psychiatry, 33 (2):270-6
SCHVEHLA TJ, MANDOKI MW,
SUMNER GS (1994) Clonidine therapy for comorbid attention deficit hyperactivity
disorder and conduct disorder: preliminary findings in a children's inpatient
unit. South Med, J 87 (7):692-5
SONI P, WEINTRAUB AL (1992)
Buspirone-associated mental status changes. Journal of the American Academy
of Child and Adolescent Psychiatry, 31 (6):1098-9
STANISLAV SW, FABRE T,
CRISMON ML, Childs A (1994) Buspirone's efficacy in organic-induced aggression.
Journal of Clinical Psychopharmacology, 14 (2):126-30
TRENT F, NAKAMURA S, TEPPER
JM (1991) Amphetamine exerts anomalous effects on dopaminergic neurons in
neonatal rats in vivo. European Journal of Pharmacology, 204 (3):265-72
WARNEKE L (1990)
Psychostimulants in psychiatry. Canadian Journal of Psychiatry, 35 (1):3-10
YOUNG ZO (1992) Buspirone in
children with attention-deficit. Psychotropics, 12 (4):15
YU DS, SMITH FL, SMITH
DG, LYNESS WH (1986) Fluoxetine-induced attenuation of amphetamine
self-administration in rats. Life Sciences, 39 (15):1383-8
ZWIER KJ, RAO U (1994)
Buspirone use in an adolescent with social phobia and mixed personality
disorder (cluster A type). Journal of the American Academy
of Child and Adolescent Psychiatry, 33 (7):1007-11
Professor Vernon M.
Neppe
Director, Pacific
Neuropsychiatric Institute, Seattle, WA,
USA;
Adjunct Professor of
Psychiatry and Human Behaviour, St Louis University School of Medicine, St
Louis, MO, USA.
Zora O. Young
MD
Department of Psychiatry and
Behavioral Science, University of
Nevada, Reno, Nevada,
USA
Correspondence:
Vernon M. Neppe, MD, PhD, FRSSAf,
FFPsych, MMed, FRCPC, DPM,
MB, BCh, Dip
ABPN
Director
Pacific Neuropsychiatric
Institute
Seattle Healing Arts Center, Suite 353
6300 Ninth Ave NE
Seattle, WA 98115,
USA
See Also: The serotonin 1A neuromodulation of aggression
